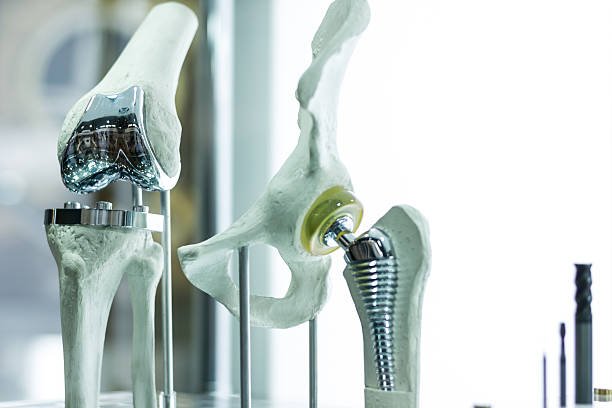
Quality assurance is the backbone of orthopaedic implant manufacturing. Since implants remain inside the body for years, every component must undergo rigorous testing, documentation, and validation. A single manufacturing inconsistency can compromise patient safety.
ISO 13485:2016 compliance ensures that a manufacturer maintains strict quality management systems, including traceability, sterile manufacturing, and risk mitigation. Cleanroom environments prevent contamination, while mechanical testing ensures the implant’s strength, fatigue resistance, and durability.
JK Digital NeoBone follows a strong quality pathway that includes design verification, raw material inspection, machining accuracy, final finishing, and sterilization validation. The company also maintains strict documentation and traceability systems, which are essential for regulatory compliance and OEM partnerships.
In orthopaedics, quality is not just a standard—it is a responsibility. High-quality manufacturing ensures safety, reduces revision surgeries, and gives surgeons and patients confidence in the product.